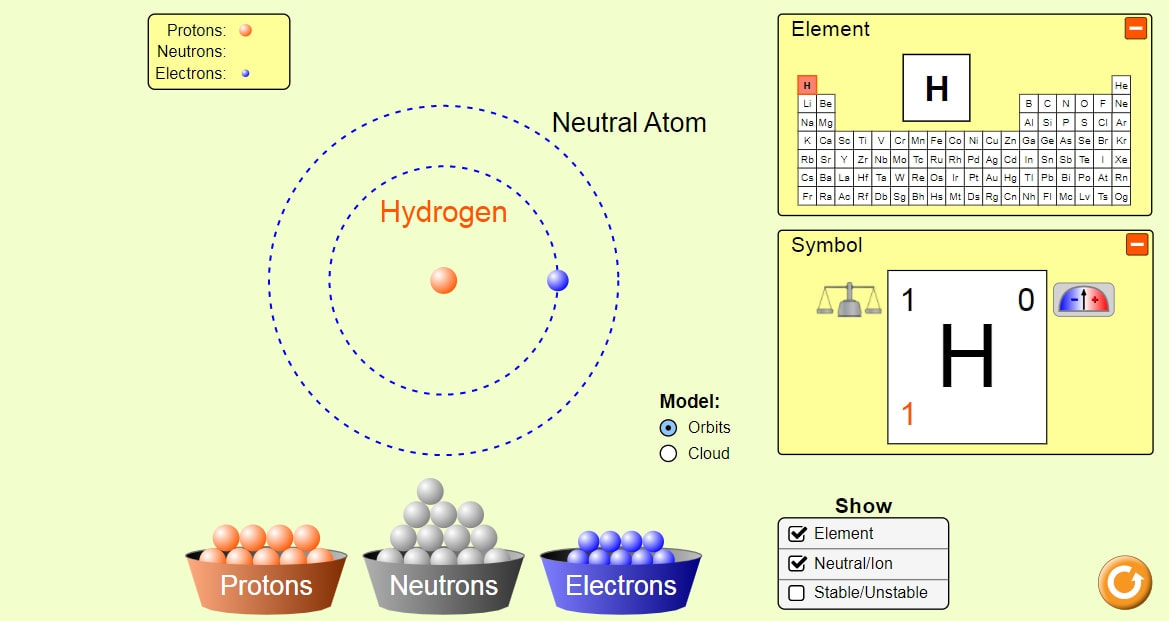
So, much like opposite ends of a magnet, protons and electrons are attracted to each other. A fundamental rule is that particles with the same charge are repulsed from each other, while particles with opposite charges are attracted to each other. Neutrons, on the other hand, don't have a charge. Two of the subatomic particles have electrical charges: protons have a positive charge while electrons have a negative charge. 
There are three subatomic particles: protons, neutrons and electrons. 
Given that these particles make up atoms, they are often referred to as subatomic particles. While its name originally referred to a particle that couldn't be divided any more-the smallest thing possible-we now know that each atom is generally made up of smaller particles. Anything that has a mass-in other words, anything that occupies space-is composed of atoms. The atom is considered the basic building block of matter. Printable Version The Nuclear Regulatory Commission's Science 101: What is an Atom?
0 Comments
Leave a Reply. |

 RSS Feed
RSS Feed
